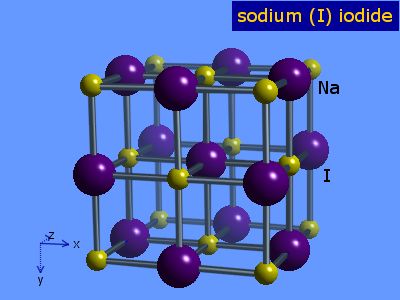
Sodium iodide

The table shows element percentages for NaI (sodium iodide).
| Element | % |
|---|
| I | 84.66 |
| Na | 15.34 |
Isotope pattern for NaI
The chart below shows the calculated isotope pattern for the formula NaI with the most intense ion set to 100%.
References
The data on these compounds pages are assembled and adapted from the primary literature and several other sources including the following.
- R.T. Sanderson in Chemical Periodicity, Reinhold, New York, USA, 1960.
- N.N. Greenwood and A. Earnshaw in Chemistry of the Elements, 2nd edition, Butterworth, UK, 1997.
- F.A. Cotton, G. Wilkinson, C.A. Murillo, and M. Bochmann, in Advanced Inorganic Chemistry, John Wiley & Sons, 1999.
- A.F. Trotman-Dickenson, (ed.) in Comprehensive Inorganic Chemistry, Pergamon, Oxford, UK, 1973.
- R.W.G. Wyckoff, in Crystal Structures, volume 1, Interscience, John Wiley & Sons, 1963.
- A.R.West in Basic solid state chemistry Chemistry, John Wiley & Sons, 1999.
- A.F. Wells in Structural inorganic chemistry, 4th edition, Oxford, UK, 1975.
- J.D.H. Donnay, (ed.) in Crystal data determinative tables, ACA monograph number 5, American Crystallographic Association, USA, 1963.
- D.R. Lide, (ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 77th edition, 1996.
- J.W. Mellor in A comprehensive treatise on inorganic and theoretical chemistry, volumes 1-16, Longmans, London, UK, 1922-1937.
- J.E. Macintyre (ed.) in Dictionary of inorganic compounds, volumes 1-3, Chapman & Hall, London, UK, 1992.
Explore periodic propertes from these links
- Abundances
- Atomic numbers
- Atomic spectra
- Atomic weights
- Block in periodic table
- Boiling point
- Bond enthalpies
- Bond length in element
- Bulk modulus
- CAS Registry ID
- Classification
- Colour
- Covalent radii
- Critical temperature
- Crystal structure
- Density of solid
- Description
- Discovery
- Effective nucl. charges
- Electron bind. energies
- Electrical resistivity
- Electron affinity
- Electron shell structure
- Electronegativity
- Electronic configuration
- Enthalpy: atomization
- Enthalpy of fusion
- Expansion coefficient
- Enthalpy: vaporization
- Geology
- Group Name
- Group numbers
- Hardness
- Health hazards
- History of the element
- Hydration enthalpies
- Hydrolysis constants
- Ionization energies
- Isolation
- Isotopes
- Lattice energies
- Liquid Range
- Mass in human
- Meaning of name
- Melting points
- Molar volume
- Names and symbols
- NMR
- Orbital_properties
- Oxidation numbers
- Period numbers
- Poisson's ratio
- Ionic radii
- Radii: atomic
- Radii - ionic, Pauling
- Radii - metallic (12)
- Radii - valence orbital
- Radii - Van der Waals
- Reactions of elements
- Reduction potentials
- Reflectivity
- Refractive index
- Rigidity modulus
- Standard state
- Superconductivity
- Term symbol
- Thermal conductivity
- Thermodynamics
- Uses
- Velocity of sound
- X-ray mass abs. coeff.
- Young's modulus
- Key properties
- Electronegativity
- Melting point
- Density
- Molar volume
- Ionization energy (1st)
- Covalent radius
- Metallic radius
- Abundance: earth's crust



